Serotonin was long thought to be an exciting chemical involved in the pathways of reward processing, happiness and mood regulation[1]. Though, recent studies bring to light the complexities of this stimulant, and how little we really know of how it works. Certainly it is an important neurochemical and it can help us to understand resilience across the 6 domains.
This article will show us some of the good and bad of the highs (yes, too much serotonin can also be unhelpful) and lows we feel through serotonin in our brain specifically, how SSRIs work, as well as how it makes us feel – including physical and mental side effects.
Learn how to regulate your serotonin level and build resilience. Get the Driven app.
What is Serotonin?
Serotonin is a monoamine neurotransmitter also called 5-hydroxytyptamine – or 5-HT. It is derived from the essential amino acid L-tryptophan – ‘essential’ meaning the body can’t create it itself, therefore we need to obtain our 5-HT from the foods we eat. Serotonin is generally involved in the body’s sleep wake cycles, appetite and reward centre[2]. Although there are 7 different families of serotonin receptors and 14 sub-types found all throughout the body where serotonin can produce adverse effects[3]. We won’t delve into all of these, though we will explore its involvement in the reward centre and how it makes us feel good when coupled with dopamine. Our main focus is serotonin, though dopamine is closely linked.
Where Do You Find Serotonin?
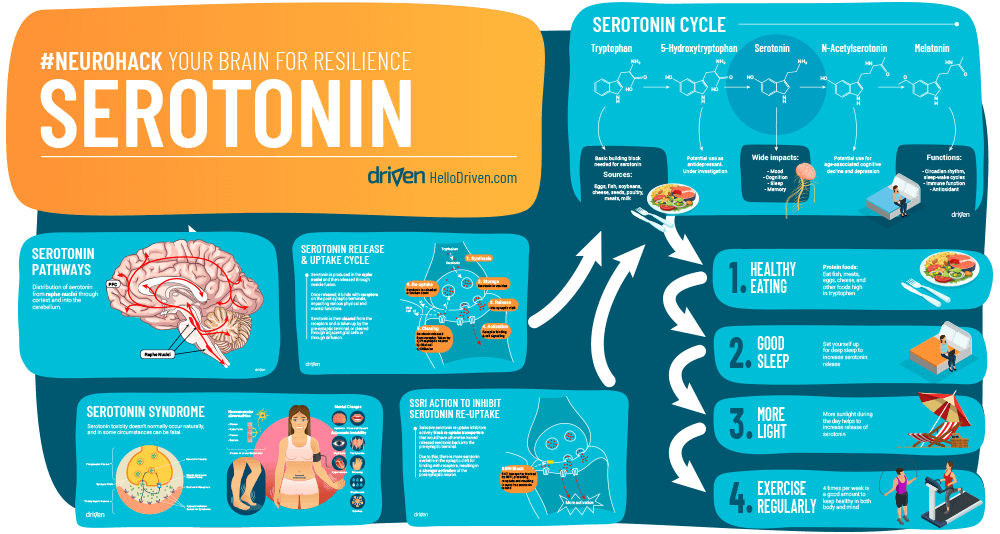
There is a lot of serotonin stored in the enterochromaffin cells found in the tissue around the gastrointestinal tract (GIT)[3]. In the gut about 90% of it is synthesised and stored while some is also stored in and travels in blood platelets. The other 10% is found in the brain stem where it is produced in the raphe nuclei and stored there in vesicles as it awaits release[4].
From the brain stem, there are over 20 known serotonergic pathways all throughout the brain[5] travelling to and through the following neural structures:
- Accumbens (ACN)
- Hypothalamus (HYP)
- Substantia Nigra (SN)
- Ventral Pallidum
- Substantia Nigra
- Ventral Tegmental Area
- Vagus Nerve
- Cerebellum
- Cortex
- Frontal Cortex
- Dorsal Vagal Complex
- Hypoglossal Nerve
- Choroid Plexus
- Olfactory Tubercle
- Limbic System
The last one is important as its effects in the limbic system means it has a large role in anxiety, depression and mood regulation.
Well, How Is Serotonin Made and Where Do We Get It From?
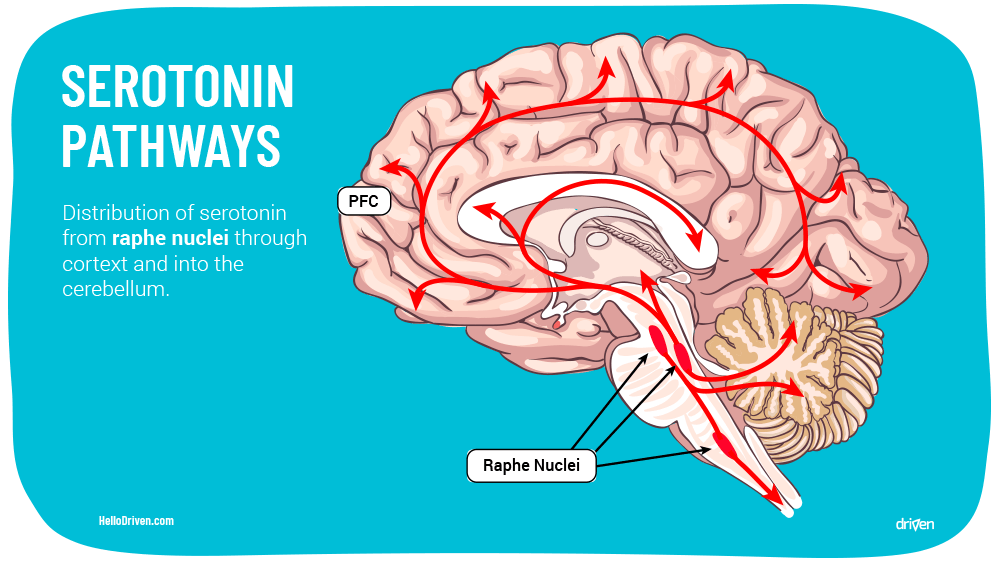
The body has no way to naturally produce serotonin, so therefore it is synthesised through amino acids consumed. 5-HT is derived from the amino acid L-Tryptophan through a process of hydration via the enzyme L-tryptophan hydroxylase and then naturally undergoes a decarboxylation process (the body forms carbon dioxide and water) to create serotonin[4].
L-tryptophan is found naturally occurring in many nuts, cheeses, fish and healthy whole foods. Because of the presence of the basic building blocks, these are sometimes called serotonin foods[6]. Perhaps this role in assisting synthesis of serotonin is why a healthy diet has been shown to reduce stress, anxiety and depression[7]. Unfortunately there is no such thing as serotonin supplements, and since serotonin can’t cross the blood brain barrier, simply sticking to a healthy diet is important to ensure the brain is in the best position to synthesise serotonin.
Physical and Mental Effects of 5-HT
With so many serotonergic pathways, it comes to no surprise that 5-HT has many physical and mental effects in the body. Let’s consider the effects of various types of serotonin dysregulation.
Physical effects of serotonin deficiency:
- Disrupted sleeping pattern (possible oversleeping) [8]
- Low appetite, craving of high sugar and fatty foods
- Low bone density
- Fatigue
- Nausea
- Chronic pain (joint, limb, back) [10]
Social effects – Because of its ‘reward’ processing and how it lifts mood (and because of the close link to dopamine), low serotonin can cause us to feel:
- High anxiety towards group exclusion (this in turn can develop a social ‘ladder’ – a competitiveness regarding friendship and reward) [11]
- Impaired learning and imitation learning ability (stress and anxiety shown to impair frontal and parietal mirror neuron system) [12]
- Impaired social bonding (serotonin genes are also linked to autism and Asperger’s) [13]
- Slow assessment of social interaction
Fear and Anxiety – Together with dopamine, we see various states that lead to feelings of stress and anxiety:
- Increased serotonin alongside decreased dopamine leads to high levels of stress and anxiety
- Decrease serotonin cause increase fear and decrease cognitive function (such as the ability to evaluate situations)
What we learn from this is that serotonin is really about achieving the right balance of receptors and serotonin production. This regulation is what allows the brain to relieve anxiety and maintain a helpful mood [14].
How serotonin can influence mood regulation and depression [4]:
- Decreased serotonin can cause an increase in impulsive behaviour (alcoholism, over-eating, destructive ‘short burst’ serotonin release, drug use [15]) due to ‘craving’ for serotonin release
- Decreased serotonin combined with decrease dopamine can result in depression and suicidal behaviour, as well as increased aggression and large shifts in mood
From this, we see that serotonin is closely involved with both depression and anxiety. Low serotonin can instigate depression coupled with anxiety [16], but also an excess of serotonin with a lack of dopamine can cause high levels of anxiety and fear.
Still, serotonin’s role isn’t fully understood in depression and anxiety. Therefore it’s quite interesting that SSRIs work based on these hypotheses and we’re still not entirely sure why they work.
Serotonin Cycle and Update
As we take a closer look at SSRI’s next, it’s helpful to understand the path that serotonin travels from the neuron releasing serotonin, and the neurons that serotonin binds with. This allows us later to see how SSRI’s is thought to function
Selective Serotonin Reuptake Inhibitors – SSRI’s
SSRI’s these days are very commonly prescribed for depression and anxiety with antidepressant and SSRI prescriptions rising continuously over the past 40 years [17].
So what are SSRI’s? Selective Serotonin Reuptake Inhibitors are a type of anti-depressant and anti-anxiety drugs designed to have a higher affinity with SERT (Serotonin transporter) than serotonin itself, with an increased chance to selectively block the presynaptic serotonin transport proteins in the presynaptic neural membrane. It is still not fully understood how this mechanism fully works to promote anti-depressant effects.
Background Check – Serotonin Release & Function
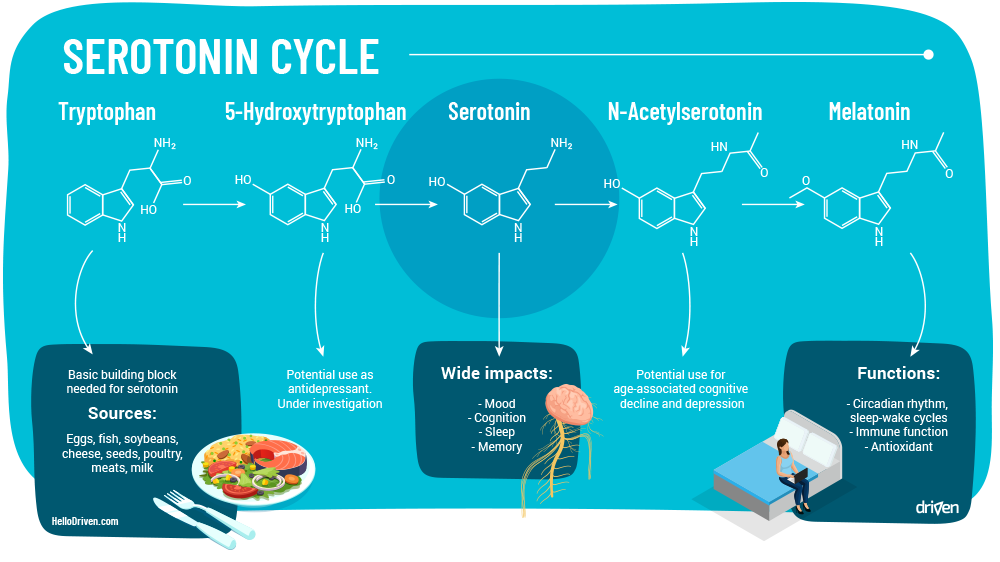
When the Raphe Nuclei nerves receive an appropriate electrical impulse, serotonin is released from vesicles stored in the pre-synaptic nerve ending. Serotonin released in synapse binds with receptor sites triggering an emotional experience until exterior stimulus has stopped.
Normally, serotonin is released readily, though due to abnormalities in external or internal stimuli (gene expression, trauma, stress, life events) there may be deficiencies. A theory as to why this happens is due to large amounts of cortisol release from stress is shown to increase the expression of SLC6A4, the gene responsible for the regulation of SERT associated with the uptake of serotonin [19].
The term deficiencies does not imply that serotonin isn’t still being released at all, but rather that it isn’t spending as much time binding to receptor sites as it should and instead is being metabolised (reuptake) faster than desired.
This can be due to a number of things, but usually via either a lack of serotonin, an increase in serotonin transporter (SERT) or an increase in monoamine oxidase (specifically MAOB – Monoamine oxidase B, the molecule that metabolises serotonin into melatonin).
This is where SSRI’s are theorised to work – we’ll take fluoxetine as an example – the structure is made to have a high affinity (binds well) with SERT, the enzyme that transports free serotonin off the receptor sites back into the neuron. Fluoxetine competes with serotonin to bind with SERT, which is why we call this molecule a ‘competitive agonist’. Here SSRI’s have varying degrees of selectivity for other monoamine transporters, hence the name ‘selective serotonin reuptake inhibitor’ (this is also why monoamine oxidase inhibitors are also theorised to treat depression and anxiety).
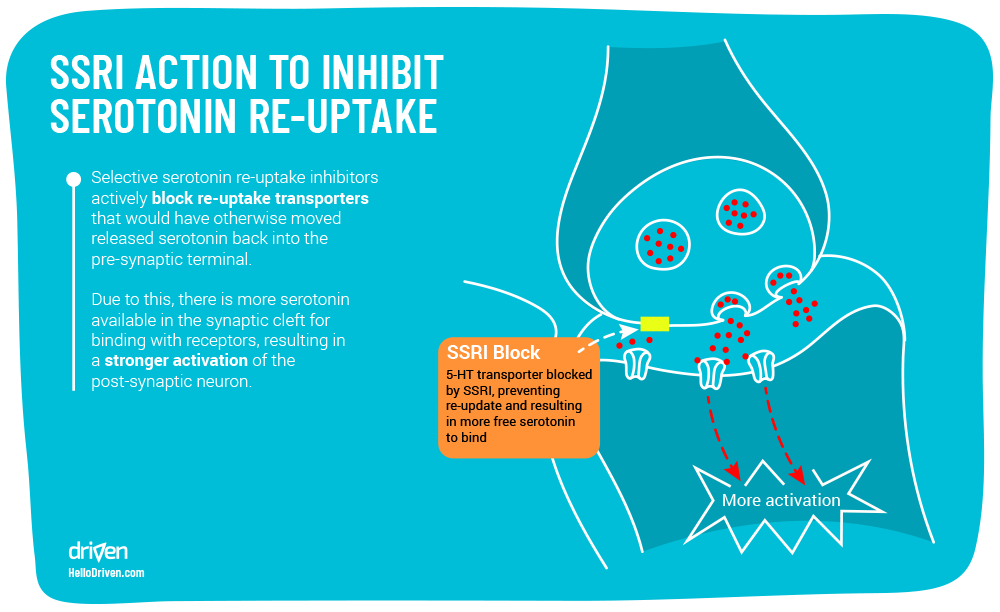
SSRIs have been shown to relieve physical symptoms of depression and anxiety in people, though there is some controversy around their performance vs placebo[23]. Considering potential side effects of long term use, there certainly is cause for care to be taken in its application.
The Side Effects of SSRI’s
- Dry mouth
- Sweating
- Headaches
- Sedation
- Dizziness
- Nausea
- Somnolence
- Insomnia
- Diarrhoea
- Impotence
Continued use of heavy doses means additional work for the liver, which could potentially cause permanently damage to its performance [22]. The true value of SSRI’s is still under debate. Indeed, multiple placebo studies [23] show that only 43% of trials showed significant benefit of drug over placebo. One of the classic precautions of SSRI’s is to not prescribe it to patients with a suicide risk, as SSRI’s may actually increase the risk of suicidality in some cases, as well as hasten the onset or worsen the course of other serious illnesses [24].
Changing pathways
SSRI’s therefore may be helpful in providing a window for treatment through talking therapies. Treatment here enables neuroplasticity to make new healthy neural connections that ideally produce more constructive thought patterns.
While SSRIs serve to increase serotonin availability, making new and healthy connections in the brain will be easier due to serotonin’s role in synapse formation. Here serotonin regulates the production of cell adhesion in the brain, which is the main component in the plasticity of a developing brain[25].
With this added help from SSRIs, CBT (cognitive behavioural therapy) has been shown to decrease symptoms and have even a greater efficacy in solving psychological disorders than SSRIs alone [26], Hoffman and Smits commented that “CBT is a reliable first line approach for treatment of anxiety and depression” [27].
Serotonin Syndrome – What Happens When Your Serotonin Levels Are Too High?
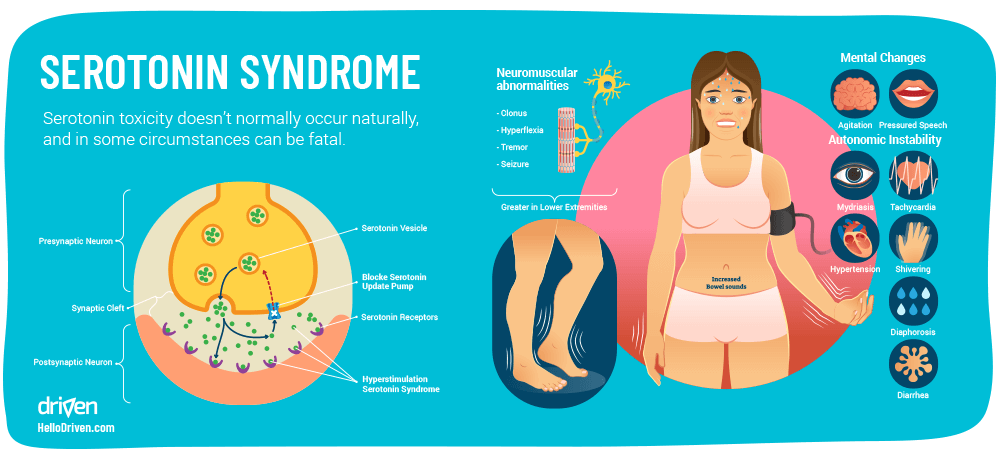
Serotonin Syndrome (otherwise known as serotonin toxicity) doesn’t usually occur naturally, but in the case of depression and anxiety and the rising use of antidepressants and SSRIs, it’s helpful to explore this further.
Serotonin Syndrome is caused by an excessive amount of serotonin in the body which becomes toxic to the CNS, to the extent that it can be potentially fatal. A case study by the American Association of Poison Control Centers Toxic Exposure Surveillance System studied 8,187 people that were diagnosed as having serotonin toxicity due to selective serotonin reuptake inhibitors (SSRIs) in 2004. Out of these people, 103 deaths occurred [28].
The mechanism of symptoms is thought to be caused by an over activation of the 5-HT1A and 5-HT2A receptors. Via this overactivation, possible frequent large mental status changes, neuromuscular hyperactivity and autonomic hyperactivity can occur – from these, possible serotonin syndrome symptoms can arise, such as: [28]
- Increased anxiety
- Suicidal thoughts
- Agitation
- Disorientation
- Restlessness
- Tremors
- Seizures
- Muscle rigidity
- Coma
It is uncommon that these arise naturally. Rather, these may arise when using larger doses SSRIs, MAOIs, TCAs and other reuptake inhibitors or metabolite blockers. Note that it is especially seen in cases of new anti-depressants coupled with existing mood stabilising drugs to form adverse drug interactions with SSRI’s[29].
Serotonin Syndrome Treatment
Treating serotonin syndrome is often about finding the drug responsible for the symptoms (most of the times the newly prescribed drug). Although due to half-lives and metabolites, symptoms can persist for up to 2 weeks, depending on the type of drug, even with continued treatment [21].
Some main treatments to stop physical symptoms of serotonin syndrome include various benzodiazepines, such as Valium, which reduces muscle agitation and stiffness/seizures, as well as serotonin blocking drugs e.g. Periactin[30].
This suggests that drugs affecting mechanisms involving serotonin in the CNS should be prescribed with care, while being vigilant of potential drug interactions and symptoms of serotonin syndrome.
Why The Long Face, is it From Your Parents?
Since serotonin was discovered in 1948 [1] and linked with depression and anxiety [31] it has been linked to be caused by a genetic predisposition [11]. In a family based study, it was shown that SERT (5-HTT) was closely linked to neuroticism and impulsive behaviour – through neural imaging (fMRI) it was shown that the single gene; SLC6A4 was responsible for the regulation of production for SERT, linked in the polymorphic region of the short chain 5HTTLPR [32].
This study suggests that the short chain variant of the 5-HTT gene is responsible for the production of the mRNA of 5-HTT, meaning that it is directly responsible for less 5-HTT in the pre-synaptic terminal – therefore resulting in higher levels of serotonin in the synaptic cleft, resulting in high levels of anxiety.
A hospital clinical study in Germany of family and twin studies calculated a genetic heritability of 31.6%, with the same predisposition across sexes. A number of imaging studies reported that presence of the 5-HTTLPR short variant is associated with greater activation to emotional than neutral stimuli in limbic and cortical brain regions [33], especially in the amygdala, which is responsible for the expression of fear and aggression as well as species-specific defensive behaviour, and it plays a role in the formation and retrieval of emotional and fear-related memories [34]. This may indicate an increase propensity to the formation of PTSD.
Increased activation in our limbic system without knowledge of techniques or situational awareness can cause many physical and unwanted effects, such as dizziness, tightness in chest, palpitations, shortness of breath etc [35]. These may be recognised as fight-or-flight responses in stressful situations. This reaction was seen to be more prevalent in people with a higher presence of short chain 5-HTTLPRs when each physiological response was tested against angry emotion in pictures.
How To Increase Serotonin, Naturally
What does all this mean for our collective serotonin regulation? There are numerous natural ways to help your body produce more serotonin in your system as well as dopamine levels and overall feeling of wellbeing. Here are four simple ways to help increase and regulate serotonin.

1. Healthy Eating
Foods rich in protein containing essential amino acids have been shown to boost levels of L-tryptophan in plasma, promoting serotonin synthesis in the CNS which may elevate mood, particularly if there was a lack in L-tryptophan affecting serotonin production.
Eating late at night can also affect serotonin levels before bed – a spike in serotonin before sleep can cause sleeplessness, which can also negatively affect your natural serotonin levels, which brings us to the next point:

2. Good Sleep
Sleeping longer and deeper has been shown to boost serotonin concentration in platelets around the body during waking hours – try to disconnect yourself before sleep and wind down. Alternatively, light from devices, especially blue light and visual stimulation can trigger excitatory neurons that may keep you awake for longer.

3. More Light
Oddly enough it has been shown that higher exposure to light can increase levels of serotonin in the CNS, although the evidence relating effects to serotonin is indirect. “In human post mortem brain, serotonin levels are higher for those who died in summer than in winter” [36] , and a positive correlation in a study of serotonin synthesis and hours of exposed sunlight in the day [37]. So, perhaps more exposure to sunlight during the day can increase serotonin levels!

4. Exercise Regularly
Whilst several articles and research papers show the correlation between exercise and its anti-depressant effect and raising of serotonin levels in the brain, it is not fully understood how this mechanism works [38]. One theory as to why this may work is the endorphin and monoamine hypothesis. Endorphin theory looks closely at the phenomenon of ‘Runner’s High’ – suggesting that exercise stimulates areas in which trigger a release of β-endorphins, which has a positive correlation to an increase in mood, although it still remains unclear whether elevations in plasma endorphins are linked to depression or anxiety [39].
Another theory as to why this works is the monoamine hypothesis. Basically that exercise increases the release and availability of neurotransmitters e.g. serotonin, dopamine, norepinephrine, this in turn has an antidepressant effect in the CNS ultimately increasing mood and lowering physical symptoms. [40]
Conclusion
Serotonin is certainly a complex molecule. What’s more, it ends up being all about balance in life and in the mind. In the therapeutic world there is much to think about in the extent to which SSRI’s should be applied, and whether there are more natural pathways to treat disorders that provide a healthier restoration in serotonin regulation.
Next up we’ll take a closer look at dopamine.
Until then, all the best,
Jurie
- Kahn, R.S., et al., Serotonin and anxiety revisited. Biological Psychiatry, 1988. 23(2): p. 189-208.
- Seo, D., C.J. Patrick, and P.J. Kennealy, Role of Serotonin and Dopamine System Interactions in the Neurobiology of Impulsive Aggression and its Comorbidity with other Clinical Disorders. Aggression and violent behavior, 2008. 13(5): p. 383-395.
- Charnay, Y. and L. Léger, Brain serotonergic circuitries. Dialogues in clinical neuroscience, 2010. 12(4): p. 471-487.
- Best, J., H.F. Nijhout, and M. Reed, Serotonin synthesis, release and reuptake in terminals: a mathematical model. Theoretical biology & medical modelling, 2010. 7: p. 34-34.
- Carhart-Harris, R.L. and D.J. Nutt, Serotonin and brain function: a tale of two receptors. Journal of psychopharmacology (Oxford, England), 2017. 31(9): p. 1091-1120
- Friedman, M., Analysis, Nutrition, and Health Benefits of Tryptophan. International journal of tryptophan research : IJTR, 2018. 11: p. 1178646918802282-1178646918802282.
- Murphy, M. and J.G. Mercer, Diet-regulated anxiety. International journal of endocrinology, 2013. 2013: p. 701967-701967.
- Ursin, R., Serotonin and sleep. Sleep Medicine Reviews, 2002. 6(1): p. 55-67.
- Simon, G.E., et al., An International Study of the Relation between Somatic Symptoms and Depression. New England Journal of Medicine, 1999. 341(18): p. 1329-1335.
- Trivedi, M.H., The link between depression and physical symptoms. Primary care companion to the Journal of clinical psychiatry, 2004. 6(Suppl 1): p. 12-16.
- Albert, P.R., F. Vahid-Ansari, and C. Luckhart, Serotonin-prefrontal cortical circuitry in anxiety and depression phenotypes: pivotal role of pre- and post-synaptic 5-HT1A receptor expression. Frontiers in behavioral neuroscience, 2014. 8: p. 199-199.
- Canli, T. and K.-P. Lesch, Long story short: the serotonin transporter in emotion regulation and social cognition. Nature Neuroscience, 2007. 10: p. 1103.
- Murphy, D.L. and K.-P. Lesch, Targeting the murine serotonin transporter: insights into human neurobiology. Nature Reviews Neuroscience, 2008. 9: p. 85.
- Jung Bang, S., et al., Projections and interconnections of genetically defined serotonin neurons in mice. Vol. 35. 2011. 85-96.
- Dfarhud, D., M. Malmir, and M. Khanahmadi, Happiness & Health: The Biological Factors- Systematic Review Article. Iranian journal of public health, 2014. 43(11): p. 1468-1477.
- Jacobs, B.L. and E.C. Azmitia, Structure and function of the brain serotonin system. Physiological Reviews, 1992. 72(1): p. 165-229.
- Parish, P.A., The prescribing of psychotropic drugs in general practice. The Journal of the Royal College of General Practitioners, 1971. 21(92 Suppl 4): p. 1-77.
- Moore, M., et al., Explaining the rise in antidepressant prescribing: a descriptive study using the general practice research database. BMJ (Clinical research ed.), 2009. 339: p. b3999-b3999.
- Tafet, G.E., et al., Correlation between cortisol level and serotonin uptake in patients with chronic stress and depression. Cognitive, Affective, & Behavioral Neuroscience, 2001. 1(4): p. 388-393.
- Ferguson, J.M., SSRI Antidepressant Medications: Adverse Effects and Tolerability. Primary care companion to the Journal of clinical psychiatry, 2001. 3(1): p. 22-27.
- Rosen, R.C., R.M. Lane, and M. Menza, Effects of SSRIs on Sexual Function: A Critical Review. Journal of Clinical Psychopharmacology, 1999. 19(1): p. 67-85.
- Friedrich, M.-E., et al., Drug-Induced Liver Injury during Antidepressant Treatment: Results of AMSP, a Drug Surveillance Program. The international journal of neuropsychopharmacology, 2015. 19(4): p. pyv126.
- Kirsch, I., Antidepressants and the Placebo Effect. Zeitschrift fur Psychologie, 2014. 222(3): p. 128-134.
- Marken, P.A. and J.S. Munro, Selecting a Selective Serotonin Reuptake Inhibitor: Clinically Important Distinguishing Features. Primary care companion to the Journal of clinical psychiatry, 2000. 2(6): p. 205-210.
- Lesch, K.-P. and J. Waider, Serotonin in the Modulation of Neural Plasticity and Networks: Implications for Neurodevelopmental Disorders. Neuron, 2012. 76(1): p. 175-191.
- Hofmann, S.G., et al., The Efficacy of Cognitive Behavioral Therapy: A Review of Meta-analyses. Cognitive therapy and research, 2012. 36(5): p. 427-440.
- Hofmann, S.G. and J.A.J. Smits, Cognitive-behavioral therapy for adult anxiety disorders: a meta-analysis of randomized placebo-controlled trials. The Journal of clinical psychiatry, 2008. 69(4): p. 621-632.
- Volpi-Abadie, J., A.M. Kaye, and A.D. Kaye, Serotonin syndrome. The Ochsner journal, 2013. 13(4): p. 533-540.
- Trindade, E., et al., Adverse effects associated with selective serotonin reuptake inhibitors and tricyclic antidepressants: a meta-analysis. CMAJ : Canadian Medical Association journal = journal de l’Association medicale canadienne, 1998. 159(10): p. 1245-1252.
- Frank, C., Recognition and treatment of serotonin syndrome. Canadian family physician Medecin de famille canadien, 2008. 54(7): p. 988-992.
- Lesch, K.-P., et al., Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region. Science, 1996. 274(5292): p. 1527-1531.
- Canli, T. and K.-P. Lesch, Long Story Short: The Serotonin Transporter in Emotion Regulation and Social Cognition. Vol. 10. 2007. 1103-9.
- Canli, T., et al., Additive effects of serotonin transporter and tryptophan hydroxylase-2 gene variation on neural correlates of affective processing. Biological Psychology, 2008. 79(1): p. 118-125.
- Martin, E.I., et al., The neurobiology of anxiety disorders: brain imaging, genetics, and psychoneuroendocrinology. The Psychiatric clinics of North America, 2009. 32(3): p. 549-575.
- Bystritsky, A., et al., Current diagnosis and treatment of anxiety disorders. P & T : a peer-reviewed journal for formulary management, 2013. 38(1): p. 30-57.
- Young, S.N., How to increase serotonin in the human brain without drugs. Journal of psychiatry & neuroscience : JPN, 2007. 32(6): p. 394-399.
- Lambert, G.W., et al., Effect of sunlight and season on serotonin turnover in the brain. The Lancet, 2002. 360(9348): p. 1840-1842.
- Craft, L.L. and F.M. Perna, The Benefits of Exercise for the Clinically Depressed. Primary care companion to the Journal of clinical psychiatry, 2004. 6(3): p. 104-111.
- Catecholamines, Dopamine, and Endorphin Levels during Extreme Exercise. New England Journal of Medicine, 1981. 305(8): p. 466-467.
- Ebert, M., R. Post, and F. Goodwin, EFFECT OF PHYSICAL ACTIVITY ON URINARY M.H.P.G. EXCRETION IN DEPRESSED PATIENTS. The Lancet, 1972. 300(7780): p. 766.